Which of the Following Correctly Describes Chemical Equilibrium
1 and 2. There is no longer a change in concentration of reactants and products over time.

Https Www Pinterest Com Explore Le Chatelier S Principle Teaching Chemistry Chemistry Lessons Chemistry Education
Which of the following statements correctly describes any chemical reaction that has reached equilibrium.
. D The rates of the forward and reverse reactions are equal. Which of the following correctly describes a reaction that has reached chemical equilibrium. A The rate of the forward reaction is equal to the rate of the reverse reaction.
A catalyst is added to the reaction system. Which of the following statements correctly describe how a system at equilibrium will respond to a change in concentration of any species that appears in the expressions for Q and K. Select all that apply.
Chemistry questions and answers. The best describe should be the concentration of reactants and products are constant. The concentrations of the reactants are equal to the concentrations of the products.
In your choices the best answer is the mass of the reactants and the mass of the products are no equal. A The concentration of products equals the concentration of reactants. The equilibrium potential describes the membrane potential where what two forces are balanced.
A At equilibrium the concentration of reactants is equal to the concentration of the products. The equilibrium constant Kc for the reaction H2g I2g 2 HIg is 55 at 425C. There is no longer a change in concentration of reactants and products over time.
C Both forward and reverse reactions have halted. B At equilibrium the forward and reverse reaction rates are equal to zero. Which of the following statements correctly describes any chemical reaction that has reached equilibrium.
Reactants are no longer reacting to form. The reactants are completely used up. Which of the following statements correctly describes a chemical system that has reached equilibrium.
The forward and reverse reactions occur at equal rates. Concentrations of products are higher than the concentrations of the reactants. C Both forward and reverse reactions have halted.
Select all that apply. Which of the following statements correctly describe a chemical system at equilibrium. Which one of the following statements correctly describes chemical equilibrium.
Which of the following correctly describes chemical equilibrium. Reactions stop only when all reactants have been converted. B Concentrations of products are higher than the concentrations of the reactants.
1 2 and 3. 3 The concentrations of reactants and products remain constant. Chemical equilibrium a condition in the course of a reversible chemical reaction in which no net change in the amounts of reactants and products occurs.
Select all that apply. Which of the following statements correctly describe a chemical system at equilibrium. About twenty-five of the ninety-two natural elements are known to be essential to life.
Which of the following statements describes a reversible reaction that has reached chemical equilibrium. The reactants are completely used up. The value for the equilibrium constant will be changed if A.
The forward and reverse reactions occur at equal rates. Forward and reverse reactions continue with no net effect on the concentrations of the reactants and products. Which of the following statements correctly describes any chemical reaction that has reached equilibrium.
A The concentrations of products and reactants are equal. Which of the following statements most correctly describe s any chemical equilibrium. Select all that apply.
The rate of the reverse reaction equals the rate of the forward reaction. A The concentrations of products and reactants are equal. There is no longer a change in concentration of reactants and products over time.
Select all that apply. B The rate of the forward reaction equals the rate of the reverse reaction. The rate of the forward reaction exceeds the rate of the reverse reaction.
1 and 3. D The rates of the forward and reverse reactions are equal. C All of the products have been converted to the reactants of the reaction.
Which of the following correctly describes all chemical equilibrium. Select all that apply Which of the following statements correctly describe a chemical system at equilibrium. B The reaction is now irreversible.
B The reaction is now irreversible. The chemical equilibrium can take place in a close system and can not be affected by catalyst and is a reversible reaction. Forward and reverse reactions have stopped so that the concentration of the reactants equals the concentration of the products.
1 The forward and reverse rates of reaction are equal. To see more answers head over to. Concentrations are given in atmospheres instead of molL.
C At equilibrium the concentrations of reactants and products do not change. The reactants are completely used up. C Both forward and reverse reactions have halted.
3 on a question which of the following statements correctly describe a chemical system at equilibrium. B A state in which the concentrations of all the reactants and products remain constant with time C A state in which the rates of the forward and reverse reactions are not equal. The forward and reverse reactions occur at equal rates.
2 No further macroscopic changes in the system are observed. A Forward and reverse reactions continue with no net effect on the concentrations of the reactants and products. D Both the forward and the reverse reactions have stopped with.
B All of the reactants have been converted to the products of the reaction. D The reaction is now irreversible. The rate of the reverse reaction exceeds the rate of the forward reaction.
Which of the following correctly describes chemical equilibrium. A A state in which the concentrations of the reactants and products are always equal. Reactants are no longer.

Chemical Equilibrium Types Problems Factors Affecting Examples

Equilibrium State And Its Characteristics Definition Examples Diagrams
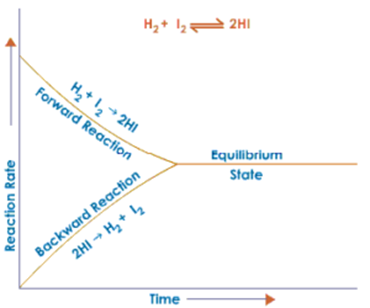
Chemical Equilibrium The Concept Characteristics Factors Affecting
What Is Chemical Equilibrum Quora
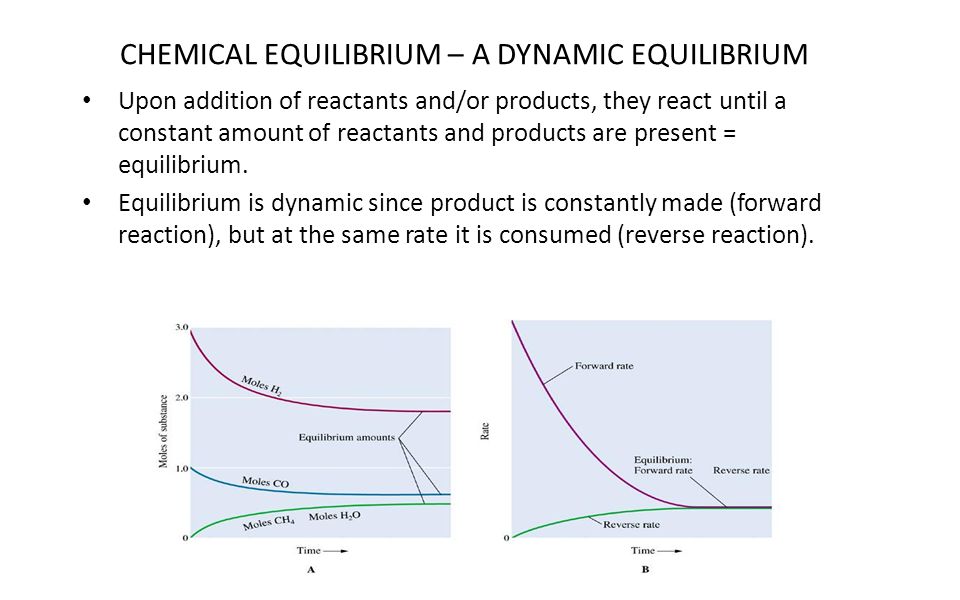
Chemical Equilibrium Overview Describing Chemical Equilibrium Chemical Equilibrium A Dynamic Equilibrium The Link To Chemical Kinetics The Equilibrium Ppt Download

Solved Ooo 15 Of 50 Which Of The Following Correctly Chegg Com
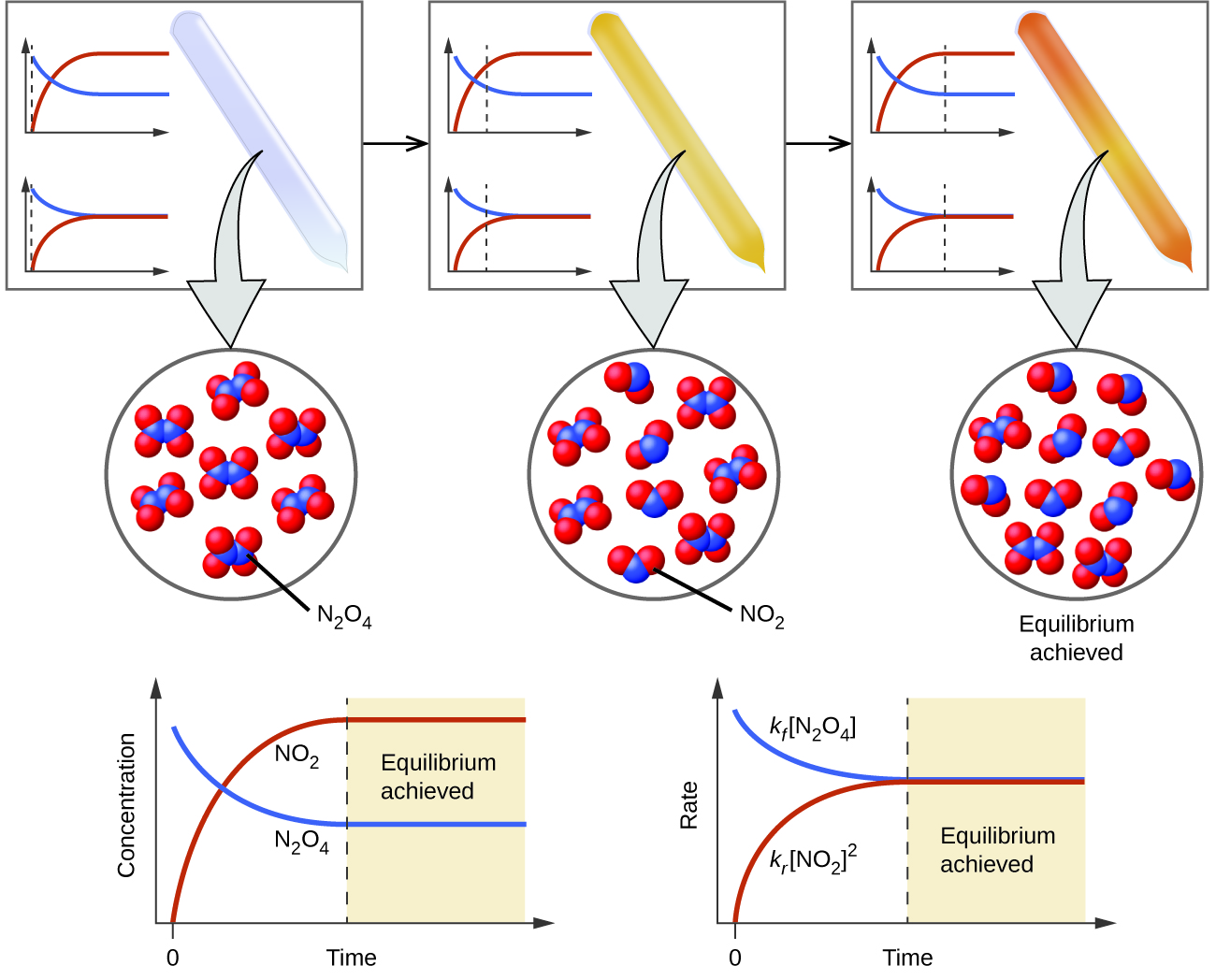
13 1 Chemical Equilibria Chemistry

8 2 Chemical Equilibrium Chemistry Libretexts

Welcome To Learnapchemistry Com Chemistry Classroom Teaching Chemistry Worksheet Template
What Is Chemical Equilibrum Quora

Chapter 4 Chemical Equilibrium Ppt Video Online Download

Welcome To Learnapchemistry Com Ap Chemistry Question Paper Ap Chem

Chemical Equilibrium Reversible Reactions Most Chemical Reactions Are Reversible What Does This Mean The Products Of A Chemical Reaction Under Certain Ppt Download

Chapter 12 Gaseous Chemical Equilibrium Ppt Video Online Download

This Guy Is Using One Of My Favorite Games To Explain Collision Theory Nick Collision Theory Theories Solubility

Chemical Equilibrium Overview Describing Chemical Equilibrium Chemical Equilibrium A Dynamic Equilibrium The Link To Chemical Kinetics The Equilibrium Ppt Download
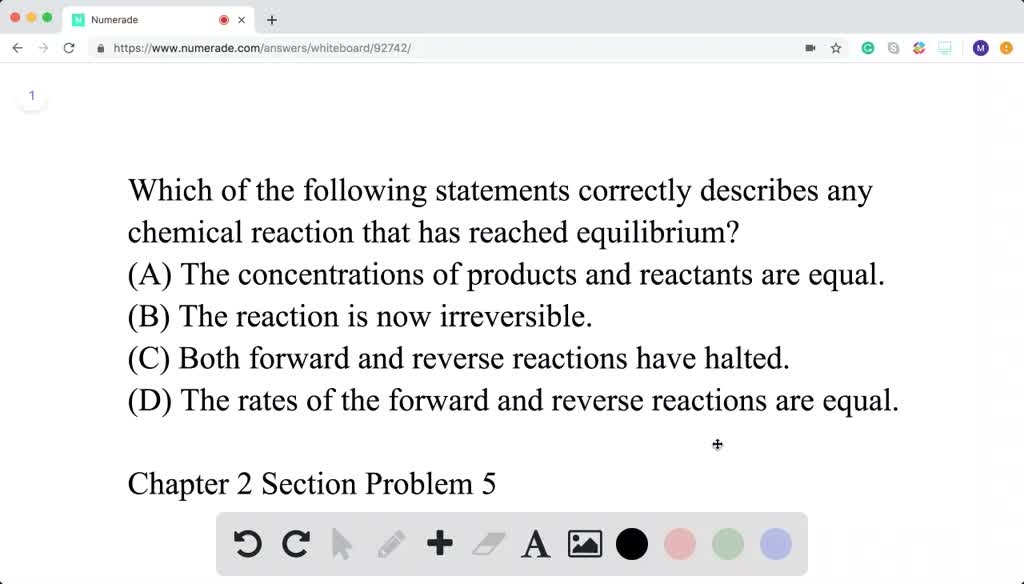
Solved Which Of The Following Statements Correctly Describes Any Chemical Reaction That Has Reached Equilibrium A The Concentrations Of Products And Reactants Are Equal B The Reaction Is Now Irreversible C Both Forward

Comments
Post a Comment